The Composition of Earth's Atmosphere
If you measured the percentage of the different gases in any fixed volume of air you would find approximately
78% nitrogen
21% oxygen
0.9% argon
The other gases in Earth's atmosphere (typically less than 0.1%) are called trace gases. Of these trace gases, water vapor is the most abundant, followed by carbon dioxide, methane and ozone.
Even though water vapor accounts for a very small portion of the atmosphere, it's contribution to weather and climate is huge. When water changes from one phase to another, such as from a liquid to a gas, energy is transferred in the atmosphere. This is an extremely important concept because the availability of water vapor essentially fuels or limits all atmospheric processes.
The Amazing Water Molecule
Water (H2O) is two hydrogen atoms attracted and bonded to one oxygen atom. Water is the only substance on Earth that exists naturally as a liquid, solid and gas. And what happens during phase changes is extraordinary.
Most of water's unique characteristics are due to it's molecular structure and the electrostatic forces within water molecules. When two hydrogen atoms combine with an oxygen atom to form a water molecule the single electron on each hydrogen is shared with the oxygen so that the hydrogen ends of the water molecule end up with a slight positive charge and the oxygen end has a slight negative charge, making water a "polar" molecule. This polarity results in an electrostatic force between neighboring water molecules with the negative and positive ends attracted to each other. This electrostatic force in water is referred to as a hydrogen bond.
Hydrogen bonds caused by electrostatic forces are the basis for many unique properties of water such as supercooled liquids, the surface tension responsible for spherical rain drops, and the fact that ice is lighter than liquid water. Think about the ramifications of this last fact - if water didn't expand when it froze into ice, lakes would freeze from the bottom up, killing all forms of life in northern lakes every winter. Unlike most substances however, water becomes less dense when it solidifies because the hydrogen bonds force water to realign into a highly ordered,crystalline form as it freezes. If this expansion didn't occur, sea ice would sink in the oceans and never melt. Life on Earth would not be possible.
The phase changes of water 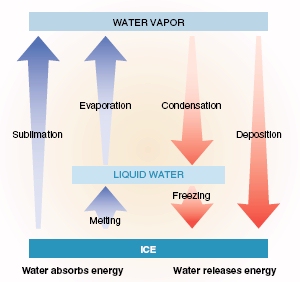
|
Because the Earth's surface is about 71% water, understanding the phase changes of water is essential for understanding the transfer of energy in the atmosphere. To break the intermolecular attractions in crystalline ice and transform it into liquid water, energy must be added, the opposite happens when ice freezes. Similarly, when water evaporates (changing from a liquid to gas) heat is absorbed and energy is removed from the atmosphere. When water vapor condenses (changing from gas to a liquid) heat is released. The daily occurrence of cloud formation results in large amounts of energy being released into the atmosphere. Atmospheric circulations routinely cause phase changes such as condensation and evaporation to occur at completely different latitudes and longitudes, redistributing heat around the planet. |
Earth's Energy Cycle
The Sun powers Earth's weather and climate with solar energy streaming through space as electromagnetic waves. Because the Sun is extremely hot, it emits radiation in short wavelengths. (Wien's Law)
All objects with a temperature emit radiation. Because Earth's surface (and atmosphere) is so much cooler than the Sun, it loses energy to space as long wave terrestrial radiation. Water vapor plays a critical role in trapping this longwave radiation in order to warm our atmosphere. Known as the Greenhouse effect, this process keeps Earth from freezing. Most of the energy that does escape from the Earth out into space travels in a narrow band of the electromagnetic spectrum called the infrared atmospheric window.
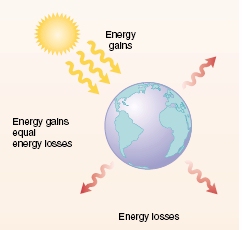
Approximately 50% of the solar energy reaching the planet passes through the atmosphere and is absorbed by the surface of the Earth, 20% is absorbed by the atmosphere and clouds and 30% is reflected back into space. The total amount of solar energy that Earth intercepts is determined by the tilt of the Earth's axis and how far the Earth is from the Sun. Although Earth is a little closer to the Sun in January than June, Northern Hemisphere temperatures are warmer in June because Earth's axis of rotation points to the Sun. The Earth's tilt, combined with it's orbit around the Sun, causes seasons.
Tropical and subtropical regions of the planet gain more solar energy than these regions lose to space by longwave radiation. Polar regions lose more energy than they receive from the Sun over any given year. This energy imbalance, with net energy losses at the poles and net energy gains in the tropics, is the driving force of weather and climate.
Earth's Energy Budget |
 |
