

The monthly average total column ozone amounts show a minimum in the southern hemisphere spring. During winter the amounts of ozone over the south pole region remain fairly constant. A decline in total column ozone is seen in September and a minimum amount of ozone is observed in October. After October the amount of ozone levels begin to increase.
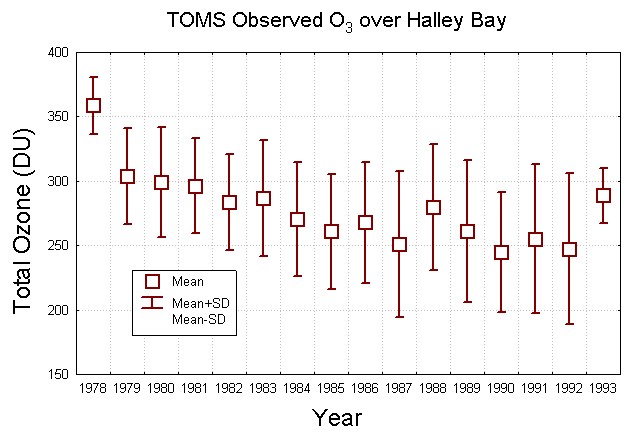
The above figure is a box-whisker diagram of TOMS observed ozone over Halley Bay, Antartica. The boxes represent the yearly averaged ozone amounts and the whiskers are plus and minus one standard deviation about the mean. TOMS observed average ozone amounts over Halley Bay Antarctica indicate that yearly average ozone amounts in the early 1990's is less than the ozone amounts observed in the early 1980's. Ground observations of ozone concentrations over Antarctica reveal a decreasing trend in total column amounts since 1955. Observations of total ozone amounts in the month of October measured over Halley Bay, Antarctica (76 degrees south latitude) demonstrate this trend.

The winter atmosphere above Antarctica is very cold. These cold temperatures result from the high altitude of the Antarctic continent and the resulting energy losses due to longwave radiation. Temperatures in the stratosphere can be less than -90C (-130F)! In addition, the cold temperatures result in a temperature gradient between the southern hemisphere polar and the midlatitudes. The temperature gradients result in pressure gradient which, in combination with the Coriolis force produces a belt of strong stratospheric winds that encircle the south pole region. The strong westerly winds, referred to as the polar vortex, prevent the transport of warm equatorial air to the polar latitudes. This isolation between the south polar regions and midlatitudes and tropical regions helps keep the stratospheric air very cold. These cold temperatures cause water vapor and some nitrogen compounds to condense and form clouds. Polar stratospheric clouds (PSCs), composed of ice and frozen nitrogen particles, form in air temperatures colder than approximately -80C or -112F. PSCs begin to form during June.
We mentioned previously that Cl is an important atom that contributes to the destruction of ozone. Chlorine atom can be bound to stable molecules such as hydrochloric acid (HCl) and chlorine nitrate (ClNO3 or ClONO2), thereby prevented chlorine from removing ozone in the stratosphere. In the winter time chemical reactions on the surface of the particles composing PSC result in chemical reactions that remove the Cl from these stable compounds. During the spring, when sunlight once again shines on the Antarctic stratosphere, the chlorine atom is freed and ozone is rapidly depleted. Destruction is so rapid over the south pole region in the southern hemisphere spring time (e.g., October) that it has been termed a "hole in the ozone layer."
There is a strong year-to-year variation in the development of the Antarctic ozone hole.

Why does the ozone hole appear more often over the south pole than the north pole? Stratospheric clouds composed of ice particles exist over Antarctica but not over the mid-latitudes or tropical regions. This is because the Arctic stratosphere does not get as cold as the air over Antarctica. As a result, while ozone depletion of 15-20% have been observed in certain regions of the Arctic stratosphere, the development of an extensive ozone hole has not been observed.
