The Electromagnetic Radiation

Electromagnetic Waves
Electromagnetic waves can be described by their wavelengths, energy, and frequency. All three describe a different property of light, yet they are related to each other mathematically. The two equations below show the relationships:
| Equation 1 | Equation 2 |
|---|---|
| f * λ = c | E = h * f |
| f = frequency | E = energy |
| λ = wavelength | h = Plank’s constant |
| c = speed of light (3 x 108 m/sec) | f = frequency |
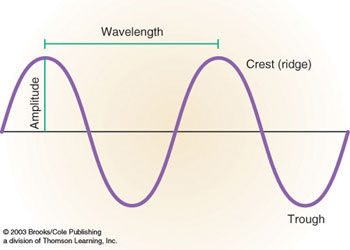
The wavelength of an electromagnetic wave is the distance between wave crests. The other
property used to describe every wave is the wave amplitude which is one half the height
from the peak of a crest to the lowest point of the wave. The distance between wave crests
is measured in terms of a micrometer or micron (unit symbol is µm).
The size of the wavelengths of radiation range from ultra-long radio waves to high-energy
gamma rays. The amount of energy in the wave increases as wavelengths
get smaller and tighter.
When radiation is measured using very sensitive instruments at extremely low light levels it is observed that the energy does not arrive continuously in waves, but rather in small, finite amounts. This is the particle view of radiation. Particles of light are called photons. Radiation can also be thought of as a collection of photons, tiny but discrete packets of energy traveling at the speed of light. Low-energy photons, like radio photons, tend to behave more like waves, while higher energy photons (i.e. X-rays) behave more like particles. So even though X-rays and Gamma rays are part of the electromagnetic spectrum, scientists generally refer to them by describing their energies rather than their wavelengths.
In satellite meteorology, we are concerned with radiant energy with wavelengths between 0.1
and 100 µm. The Sun, for example, emits most of its radiant energy with a wavelength between
0.2 to 4 µm. Solar energy, frequently referred to as shortwave radiation (remember where
it is on the spectrum), includes ultraviolet, visible, and near-infrared radiation.
| 3 / 7 |





